It’s an odorless, tasteless, and colourless gas. 
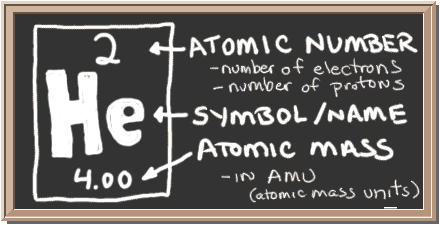
Helium is a chemical element that exists as an inert gas or noble gas. Atomic mass is also defined as the total number of neutrons and protons present in an atom. 
The mass of an atom in a given substance is known as its atomic mass. We may compute the mass of helium gas in kg/mol using the formula above. The mass of helium gas must be calculated. We can compute the mass of helium gas using the method above. We have supplied the moles of helium gas and the atomic mass of helium gas, which are one and four u respectively, in the following question. Remember that the relationship between these quantities is that moles equals mass divided by molar mass. The name for the element comes from the Greek god of the Sun, Helios.Answer: The mass of 1 mole of helium gas can be estimated using the mass, moles, and atomic mass of the helium gas relationship. Evidence of helium's existence came from the observation of a yellow spectral line from the sun.Though helium is non-toxic, breathing it can result in asphyxiation due to oxygen deprivation. Although many people think inhaling helium makes a voice sound higher, it doesn't actually alter the pitch. Inhaling helium temporarily changes the sound of a person's voice.In other words, helium is found as single atoms of the element. Helium is monatomic under standard conditions.Helium is the second least reactive noble gas (after neon). It is considered the real gas that most closely approximates the behavior of an ideal gas.Most helium is obtained by extracting it from natural gas. Its uses include helium party balloons, as a protective inert atmosphere for chemistry storage and reactions, and for cooling superconducting magnets for NMR spectrometers and MRI machines.Under high pressure, disodium helide (HeNa 2), the clathrate-like titanate La 2/3-xLi 3xTiO 3He, the silicate crystobalite He II (SiO 2He), dihelium arsenolite (AsO 6♲He), and NeHe 2 may exist. In order to bond it to another element, it would need to be ionized or pressurized. Helium is colorless, odorless, tasteless, non-toxic, and inert. Of all the elements, helium is the least reactive, so it does not form compounds under ordinary conditions.Some scientists are concerned we might one day run out of helium or at least make it prohibitively expensive to isolate. Helium is the second-most abundant element in the universe (after hydrogen), though it is much less common on Earth. On Earth, the element is considered a nonrenewable resource. Helium does not form compounds with other elements, while the free atom is light enough to escape Earth's gravity and bleed out through the atmosphere.This is because the most common isotope of hydrogen has one proton and no neutrons, while each helium atom typically has two neutrons as well as two protons. Even though hydrogen typically exists as a diatomic gas, consisting of two atoms bonded together, a single atom of helium has a higher density value. Helium is the second-lightest element. The lightest element or one with the lowest density is hydrogen.Helium has the lowest melting point and boiling point of the elements, so it only exists as a gas, except under extreme conditions. At normal pressure, helium is a liquid at absolute zero. It must be pressurized to become solid.It is energetically favorable for each helium atom to have 2 electrons, which gives it a stable electron shell. The atomic number of helium is 2, meaning each atom of helium has two protons. The most abundant isotope of the element has 2 neutrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
